.png)
Working together for smarter winter maintenance and cleaner Pennsylvania waters
Road Salt Pollution: Its Effects and Actions that can be Taken to Reduce its Usage
What happens to all that salt they put on the roads and parking lots in the winter? It just gets washed away by the next rainstorm. Right? Well actually, no. Unfortunately, pollution from excessive use of road salt is having a major impact on streams and aquifers (groundwater) in our region as well as contaminating drinking water, soil, and causing damage to infrastructure.
Statistics show that the amount of road salt being applied has doubled since 1975 (see graph). This is driven in part by increased development, resulting in more roads and parking lots that need de-icing. It is also caused by changing expectations about how much salt should be used and concerns over liability for slips and falls. All these factors result in excessive use of salt products, which is way beyond what is necessary to efficiently clear ice.

Carey Institute

Chloride loads measured in Policy-Porcupine New Hampshire watershed region (Burak, et al., 2008)
This graph was created from research done in New Hampshire and shows the sources of chlorides in a watershed. As you can see about 50% is from parking lots which are mostly maintained by private contractors. After that, we have municipal roads with 27% and state roads with 9%. Salt piles contribute 7%. The rest is a variety of smaller sources like water softeners, food waste and atmospheric deposition. Besides the sources listed on this graph, there are other sources of chloride in Pennsylvania. It can also come from wastewater treatment plants and industrial processes, and in certain areas of the state it can come from mining and well drilling processes.
Deicing chemicals work by lowering the freezing point of water which prevents ice from forming and causes existing snow and ice to melt. The most common ones are sodium chloride (rock salt), magnesium chloride, calcium chloride, and calcium magnesium acetate. They all have slightly different properties in terms of effective temperature ranges and specific environmental impact (see chart), but even the ones labeled “safe” contain chlorides which are problematic in many ways.

Photo: Jennifer Latzgo
This chart describes the properties of various types of deicers

Table contents from: https://stormwater.pca.state.mn.us/how_salt_works_and_overview_of_deicing_chemicals
Why are these chemicals harmful? After they dissolve, they separate into anions (chloride), and cations (sodium, calcium, and magnesium). Once chloride is dissolved, it stays dissolved because it does not easily form solid deposits. It is also not biodegradable, does not readily participate in biological processes, and does not strongly adhere to mineral or soil surfaces. This means that it travels very easily and impacts groundwater, soil, lakes and wetlands. Studies indicate that approximately 45% of the chlorides applied for deicing are eventually carried to the groundwater.
Once road salt is applied, subsequent rain events wash some of it directly into streams, ponds, wetlands, and lakes as runoff. In streams, this causes large temporary spikes of salt levels as much of the salt washes downstream. These spikes can be lethal to fish, insects, and other creatures that live in the creeks.
The remaining salt gets washed onto the ground and then is transported down into the aquifers below. Once there, it gradually accumulates making the groundwater saltier and saltier. Unfortunately, any chloride that enters the groundwater will stay there for a very long time, because groundwater moves slowly and there is no mechanism for salt to be removed.

Photo: Mary Rooney
This is an issue for the streams in Pennsylvania because most of them are “gaining” streams which means that they are fed year-round, primarily by the groundwater (see image). Rain events also supply streams with water, but these only lead to temporary increases in stream level. During times when there is no rain, streams continue to flow because they are being fed by groundwater. At these times, measurement of salt in stream water can give a good indication of the degree of contamination of the groundwater.

Illustration courtesy of the Groundwater Project
Effects on Stream Ecosystems
Historically, different thresholds have been used to determine toxicity to stream wildlife. Some agencies still use the US Environmental Protection Agency level of 230 mg/l that was established in 1988. However, current research shows that a level of chloride above 50mg/l in stream water is harmful to the ecosystem.

Chloride reduces oxygen levels and interferes with osmosis. Other chemicals in deicing products are also harmful to stream wildlife.
Macroinvertebrates such as Ephemeroptera (mayflies), Plecoptera (stoneflies), and Trichoptera (caddisflies) are used as bioindicators of stream water quality. These “EPT” organisms are particularly sensitive to salinization which causes osmotic stress and makes it hard for them to maintain their internal salt balance. They play a crucial role in the food web by contributing to decomposition of organic matter as well as being an important food supply source for fish and other organisms. The graph below shows how the number of EPA organisms (Taxa) decreases as chloride levels rise.


EPA National Aquatic Resource Survey (NARS) and Stroud Water Research Center
Other life in streams, lakes, and wetlands is also negatively affected by salt. This includes zooplankton that live in ponds, lakes, and wetlands. They are important because they consume phytoplankton which can be the cause of algae blooms. Amphibians such as wood frogs and salamanders are vulnerable because they absorb salt through their skin. Also, wood frog tadpoles hatch in early spring when the salt levels are highest and can suffer deformities as well as increased mortality. Studies on fish have shown reduced rates of hatching, reduced larval development rates, and reduced growth overall.
Salt also affects aquatic plants by affecting their ability to absorb water they need through their roots.

Brook Trout: Photo Jennifer Latzgo

Lake Shohola Reservoir: Photo Percons via Pixabay
In lakes and ponds, high chloride levels can reduce oxygen levels in water and make it denser, causing it to sink to the bottom. This can result in the formation of an oxygen-deficient layer which is harmful to aquatic creatures.
Birds
For our birds, the application of road salt can be dangerous in several ways. Birds’ kidneys are less efficient than mammalian kidneys at removing sodium, and if they take in too much salt, they need to increase their water intake. This means that directly ingesting salt particles can be lethal. Sadly, dead birds have been observed on roadsides after salt has been applied in winter. Other birds have also been seen exhibiting strange behavior such as fearlessness or appearing to be weak, sick, or slow: all signs of having ingested too much salt.
Why are birds attracted to road salt? Studies have found that they are looking for grit to digest their food, and the salt particles are the same size as grit. If birds do not have access to water, which may well be the case in wintertime, they can experience toxicological effects and die after ingesting as little as one particle of salt. Additionally, being attracted to roadside areas, and having reduced fear, means that they are at high risk of being hit and killed by vehicles.
The other risk for birds is that increased salt levels interfere with the food webs that they depend on.

Pine Siskin. Photo: Beheimatfilm via Pixabay
Impacts on Soil
Road salt ends up in soil as a result of runoff, plowing, bouncing, and splashing. This effect can be as far as 10 meters from the road edge. If there is heavy traffic that grinds it into finer particles, and a lot of wind, it can be blown as far as 300 feet away.
Accumulation of sodium from sodium chloride can reduce soil permeability and fertility and increase alkalinity and density causing chemical changes and a reduction in the soil’s ability to retain water. Soil microbes are also impacted, and many studies show a reduction in their activity, biomass, and community structure.
Vegetation
Road salt can significantly harm vegetation near roadways by disrupting water uptake through the roots, leading to symptoms like browning leaves, stunted growth, wilting, leaf drop, and in severe cases, plant death; this occurs due to the salt accumulating in the soil and causing root damage. This disrupts the plant's ability to absorb necessary nutrients, causing stress and dehydration even when water is available.
Salt can cause direct contact damage when salt spray from melting snow directly damages leaves and stems, causing visible browning or burning, particularly on sensitive plants like evergreens. High levels of salt can also interfere with the uptake of essential nutrients by plants.

Grass Kill. Photo: Mary Rooney

Effects on Drinking Water Quality
Elevated levels of salt in aquifers can negatively affect our drinking water supply. Much of our drinking water comes from rivers and groundwater, both of which can contain elevated concentrations of road salt chemicals. The vast majority of road salt used is sodium chloride, which dissolves into sodium and chloride ions.
If current trends continue and sodium concentrations in water supplies continue to rise, this could pose a serious concern for people with hypertension or others who are sensitive to sodium. Importantly, conventional drinking water treatment processes do not remove dissolved road salt chemicals. Technologies capable of removing sodium and chloride, such as reverse osmosis, are extremely expensive.
Salt pollution in water significantly accelerates the corrosion of water mains and pipes, primarily due to the chloride ions in salt which function as an electrolyte and speeds up the chemical reactions that lead to metal breakdown and rusting. This can lead to the leaching of harmful metals like lead into the drinking water supply.
Homeowners who have a private water supply with a well can also be affected. Elevated levels of chloride can result in corrosion of galvanized pipes. If a well is sited near a major highway, parking lot, or road salt storage facility, there may be highly elevated levels of chloride. Here In Pennsylvania, there is no mandatory testing of well water so many people are unaware of what is in the water they are drinking.
Infrastructure
The corrosive effects of road salt on cars, trucks, bridges, and roads costs approximately $5 billion a year in the United States.
Salt causes concrete to crumble and become discolored over time. This happens when the chemical, calcium hydroxide in concrete reacts with the calcium chloride in salt to create calcium oxychloride. Concrete is porous, and when salty water seeps inside concrete, the calcium oxychloride crystals expand causing cracks and crumbling. The cracks and pores are also subject to freeze-thaw effects which are more extreme with salt solution causing further damage.
Once the salt has penetrated the porous surface, it can reach the metal reinforcements inside the concrete and corrode them, causing long-term damage.

Avinash Kumar via Pixabay

Flooring
Road salt can significantly damage flooring, primarily by scratching the surface with its abrasive crystals which dull the finish and can even corrode the material, potentially causing stains and long-term deterioration, especially on hardwood floors and other polished surfaces. The longer salt residue remains on the floor, the greater the potential for damage. Stripping and rewaxing a commercial floor can cost tens of thousands of dollars.
Photo: Jennifer Latzgo
Pets
Animals can ingest deicing chemicals if they lick their paws after walking outside or eat treated snow. This can result in stomach upset, vomiting, diarrhea, and dehydration. extreme cases, animals can experience tremors and seizures. Calcium chloride can cause mouth ulcers.
Read about how to protect your pets here:
https://www.aspca.org/news/keeping-pets-safe-during-winter-dangers-ice-melts

Photo: Jennifer Latzgo
How to Mitigate Road Salt Pollution
What homeowners can do:
-
Shovel frequently so snow does not accumulate and turn to ice.
-
Salt works best on a thin layer of ice; always shovel any excess snow/ice before applying it.
-
Using more salt does not make snow and ice melt faster; it’s not necessary to feel the crunch underfoot for the salt to be effective: one 12-ounce coffee mug of salt is sufficient for a 20-foot driveway or 10 sidewalk paving squares. The grains of salt should be about 3 inches apart as shown below in the picture on the right.
-
Only apply salt in places where it is necessary.
-
After the storm, sweep any excess salt and use it for the next time it snows.
-
You can also use alternatives to rock salt such as sand, kitty litter and bird seed.
-
If bad weather is forecast, cover small areas such as steps with a tarp to prevent snow accumulation.
-
Remember that it only takes 1 teaspoon of salt to pollute 5 gallons of water!
-
Using brine, a solution of salt and water can result in better snow/ice melting while using significantly less salt. You can make your own brine solution at home by mixing 3.5 cups of rock salt with one gallon of hot tap water, stirring until the salt is fully dissolved. Allow any gravel or sediment to settle at the bottom. Transfer the brine to a standard garden sprayer and apply it in a zigzag pattern over the desired area. Any leftover brine can be stored for future use.

I'm a paragraph. Click here to add your own text and edit me. It's easy.
Image courtesy of Izaak Walton League
Best Management Practices for Municipalities and Private Contractors
By using best management practices, it is possible for a municipality or private contractor to reduce road salt usage by 50% and save a significant amount of money on materials and labor costs while maintaining safe roads, paths, and parking lots in wintertime.
Watch the documentary “The Road Map to Road Salt Reduction” to see how this was done in New York State:
https://salt.lakegeorgeassociation.org/
Calibrating Spreaders
When granular rock salt is used, calibration of salt spreading equipment is a very low-cost Best Management Practice that can result in as much as a 50% reduction in salt usage. Each truck should be calibrated annually and after a spreader is serviced. Trucks should also be calibrated for each type of deicing material that is used. The goal of calibrating is to know how much material is put down for every setting on the truck that is used. The current PennDOT recommendation is between 100-300* pounds per lane mile, depending on conditions. A “lane-mile” refers to one lane of travel, so one two lane road would be two lanes per mile.
Hamilton Township in New Jersey calibrated all their equipment twice during the winter of 2004 to 2005. Even though there were more snow events that winter and six inches more of snow, the township used $160,000 less of salt.

Photo: Marilee Ebus
Accurate Weather Forecasting

Photo: Rita E via the Noun Project
When reliable, up-to-date forecasts—especially those targeted down to the zip-code level—are available, a municipality or contractor can better predict, plan for, and respond to changing weather conditions.
With localized forecasting, they know precisely when and where deicing and plowing are needed, rather than treating large areas unnecessarily. This leads to greater operational efficiency because crews, materials, and equipment can be scheduled and deployed much more strategically.
Hyper-local forecasting services can also provide guidance about whether deicing is necessary at all, which ratio of additives should be used when preparing brine for current conditions, and the optimal timing for applying deicer.
All of these improvements enhance safety for both the traveling public and maintenance crews by ensuring roads receive the right treatment at the right time. Ultimately, the result is significant cost savings—less wasted material, reduced overtime, and fewer emergency call-outs.
Overall, accurate and highly localized weather forecasting transforms winter operations into a more proactive, efficient, and cost-effective process.
Road Condition Information Systems


Images: Courtesy Vailala
Road condition information systems employ in-road sensors like the one in the top picture, and remote sensors which are shown in the picture below, to provide detailed real-time data about road conditions.
In-road sensors are embedded directly in the pavement and measure a combination of the current amount of deicer applied, its concentration, and temperatures.
Roadside remote sensors can identify water, ice, slush, snow, and frost. They can also determine grip.
Truck-mounted sensors allow plow drivers to monitor road temperature, in real-time, which allows the driver to adjust application.
Maintenance personnel who have road temperature information are able to make better-informed decisions about how to use resources resulting in materials, fuel and labor cost savings.
Cranberry Township, PA saved $300,000 in two years by avoiding unnecessary call outs using localized weather forecasts and road temperature sensors.
Pre-Treatment: Brining
This image shows brine stripes applied to a city street in Allentown, PA.
Brine is a solution made by mixing 23.3% salt with water. It is sprayed on roads before a storm as a pre-treatment to prevent snow and ice from bonding to the surface.
Brine can be produced on-site or purchased pre-mixed. Once applied, brine stays in place meaning there is no wastage from bounce and scatter. As snow falls, it begins working immediately by preventing the snow from bonding with the road surface — this makes plowing faster, easier, and more effective.
Using brine can significantly reduce the need for traditional rock salt—by up to 75%—leading to major savings in materials, labor, and fuel.

Photo: Jennifer Latzgo
Fayette Township in Allegheny County started using brine in 2013. The township has 100 lane miles of road and experiences about 30 snowfall events a year. Before brining, they used about 90 tons of salt during each pass of their roads. They now use only 5 – 10 tons of salt to make enough brine to treat all their roads – that’s a savings of 80 tons of salt per snow event. The overall savings are about $141,000 a year.
Anti-Icing Decision Tree

This is a decision tree prepared by PennDOT which aids employees in determining the appropriate times to apply brine.
Pre-Wetting Salt
If rock salt is used, pre-wetting it by spraying it with brine before it is spread will make it stick to the road resulting in less waste from bounce and scatter. It will also activate to melt snow and ice faster. Pre-wetting rock salt can result in a 30% reduction in the amount of salt that is needed. Michigan’s DOT found that pre-wetting resulted in cost savings of one third, and that 78% of the salt stayed within the center of the roadway.
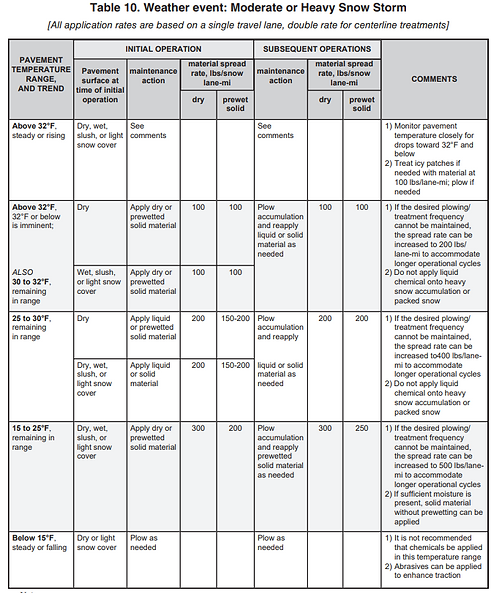
Deicing Application Rate Charts
PennDOT and other Agencies have created Application Rate charts based on weather and temperature which can be used to make informed decisions about deicing based on road conditions.
Automated Spreader Controls

Photo: Western Products
Automated spreader controls are systems in salt trucks that allow operators to program variable salt application rates so the amount of salt being applied is adjusted with the speed of the truck. They can also adjust the amount of salt being applied on curves and hills that require more salt than straight roads. Vehicle location sensors can mean that precise locations along a route can be targeted. The data provided by this type of system can allow highly informed decisions by maintenance management.
Segmented/live edge snow plows

Another way to reduce salt usage is to use segmented plows like the one in this picture. These have a blade made of short sections that are spring-loaded. This allows the blade to conform to the road surface and provides a much cleaner scrape. Because the plow conforms to the curvature of the road, a wider blade can be used. This means that fewer passes are needed to clear the roadway. Introducing these plows produced a 50% reduction in salt use in Lake George New York. Fewer roadway passes also result in significant labor and fuel savings.
Training of Staff
Staff training is another low-cost best management practice. All staff should receive the training upon hire, and then annually.
Training should include an educational component about the impacts of deicers on the environment and infrastructure, and how reducing their usage can result in significant cost savings. There should also be instruction about how deicing works and the protocols and procedures that should be used.
In Pennsylvania, PennDOT provides training to municipalities through its LTAP program: https://gis.penndot.gov/ltap/

Salt Storage
Road salt that is not stored properly can be dissolved by rain resulting in salt runoff and leaching into the ground. Salt that gets wet can also lump together making it more difficult to load and apply.
Here are some guidelines for salt pile storage:
-
All salt piles should be covered by permanent structures like the one in the picture, or kept under weighted, heavy-duty tarps.
-
They should be on an impervious surface, and the site should be level. and outside the 100-year floodplain as well as areas of localized flooding and away from stormwater facilities.
-
There should be adequate drainage controls to prevent any runoff.
-
After loading, staff should clean up any spills that occur.
-
Salt piles should be inspected and maintained regularly.

Treatment of recycling of truck wash water
When salt trucks and equipment are washed, the wash water and runoff can contain high levels of sodium, chloride, and other pollutants that enter the environment. Wash water should be captured and treated, then it can be used as salt brine for pre-wetting and anti-icing.
.png)
Photo: Courtesy of Interclean
Evaluation of performance

Throughout the season, there should be ongoing monitoring, reporting and analysis of performance.
There should also be an annual evaluation by management to determine whether objectives were met, track the deicing materials that were used and identify areas where improvements need to be made in the future.
Learn more
For more in-depth information, explore the online resources listed below:
Video: Salt Impacts on Water Quality - John Jackson, Stroud Water Research Center
Cary Institute of Ecosystem Studies: Road Salt The Problem, The Solution, and How to Get There
PA DEP & Southwestern PA Commission: Quick Resource Guide for Winter Maintenance BMP's
Salt Institute: Salt Storage Handbook
https://www.nwpa.us/uploads/1/2/9/8/129889926/salt-institute-salt-storage-handbook.pdf
United States Environmental Protection Agency: Deicing Material Application and Storage
https://www.epa.gov/system/files/documents/2021-11/bmp-deicing-material-application-and-storage.pdf